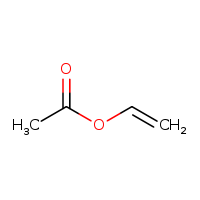
The VAM molecular formula is C4H4O2. Its CAS is 108-05-4. VAM is considered slightly toxic. The clear, colorless liquid is slightly soluble in water and its vapors are heavier than air. VAM is primarily used as a monomer in the manufacture of polyvinyl acetate and polyvinyl alcohol. Short-term exposure has resulted in eye and upper respiratory irritation.
The industrial preparation of VAM according to the Journal of Catalysis, involves the reaction of ethylene ad acetic acid with oxygen in the presence of a palladium catalyst.
Industry statistics estimate that approximately 80% of global VAM production is used to manufacture PVA and PVOH, with most of the remaining volume being used in the production of PVB, EVA copolymers and EVOH resins.